
Introducción
Understanding the world of genomic analysis is essential for advancing personalized healthcare, particularly for genetic counselors who are key in interpreting genetic data. This guide outlines the critical steps that enable these professionals to effectively utilize genomic insights, from data collection to reporting findings. As the field evolves rapidly, genetic counselors must consider how to stay at the forefront of this practice while addressing the complexities of ethical compliance and data integrity.
Define Genomic Analysis and Its Importance
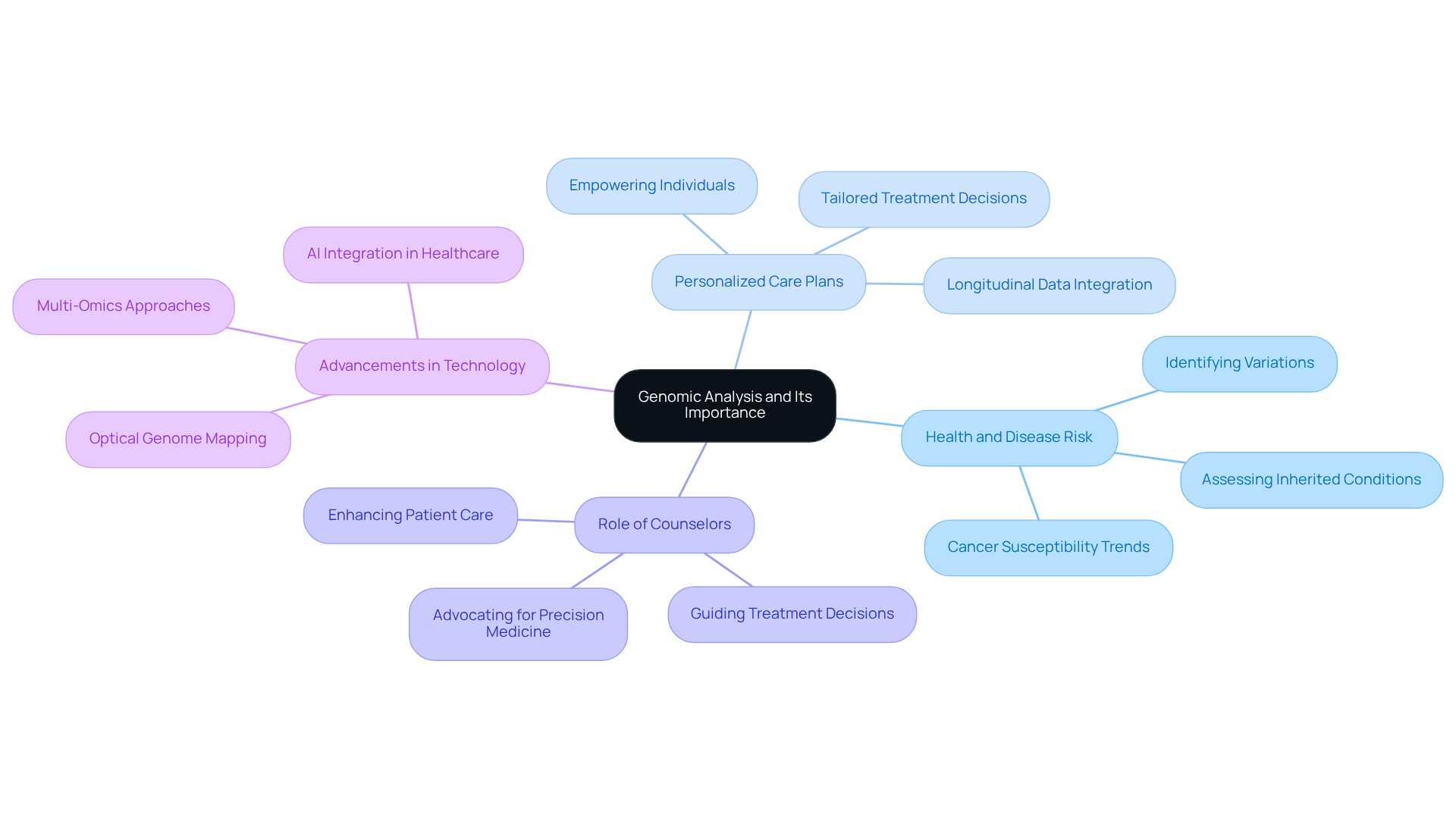
Understanding an individual’s hereditary material is crucial through genomic analysis, as it identifies variations that can significantly influence health, disease risk, and treatment responses. This foundational insight underscores the importance of genomic analysis for counselors, allowing them to create personalized care plans that are tailored to each person’s unique genetic profile.
By leveraging genetic insights, counselors can effectively assess inherited conditions, guide treatment decisions, and inform individuals about their hereditary risks. This personalized approach not only enhances patient care but also empowers individuals with knowledge about their genetic predispositions.
Recent advancements in genomic analysis, particularly the shift towards multi-omics approaches, have further refined the ability to customize healthcare. For instance, integrating genetic data with longitudinal clinical records has revealed trends in cancer susceptibility and the occurrence of rare diseases, facilitating more accurate risk evaluations.
As genomic analysis evolves, staying abreast of current trends will enable genetic counselors to play a pivotal role in advancing precision medicine. This ongoing development is essential for enhancing care and outcomes for individuals.
Collect Relevant Genomic Data
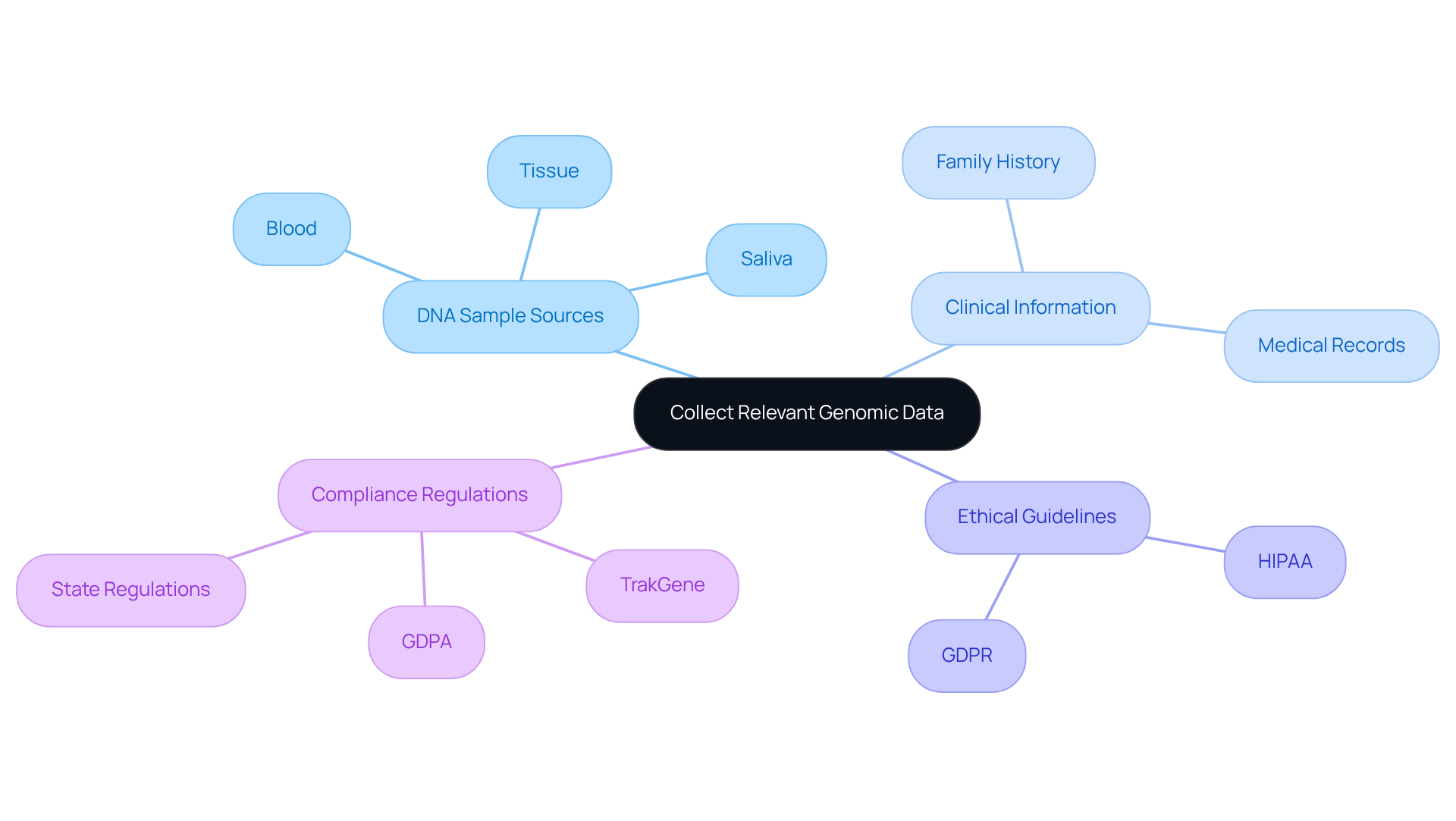
To initiate the genomic analysis process, gathering extensive genetic information is crucial. This involves obtaining DNA samples from patients, which can be sourced from blood, saliva, or tissue. Additionally, relevant clinical information, such as historia familiar and medical records, should be collected to provide context for the genetic data.
Accessing online databases and DNA repositories can significantly enhance your genomic analysis by integrating existing genetic datasets. It is imperative that all information collection practices adhere to ethical guidelines and regulatory standards, including HIPAA and GDPR, to protect patient privacy and maintain the integrity of the information.
TrakGene is dedicated to ensuring compliance with these regulations and has established an ISO27001 Information Security Management System to uphold industry standards for information security. Recent legislative efforts, such as Nebraska’s 2024 law mandating direct-to-consumer testing companies to publish privacy policies and obtain consent for information collection, highlight the critical nature of compliance in genetic practices.
Furthermore, the Bipartisan Genomic Information Protection Act (GDPA) aims to regulate the collection of hereditary information, emphasizing the necessity for adherence to ethical standards. It is also essential to be aware of the variability in state regulations regarding genetic information protection, as this landscape can significantly influence compliance and best practices in the collection of genetic information.
Perform Data Quality Checks and Cleaning
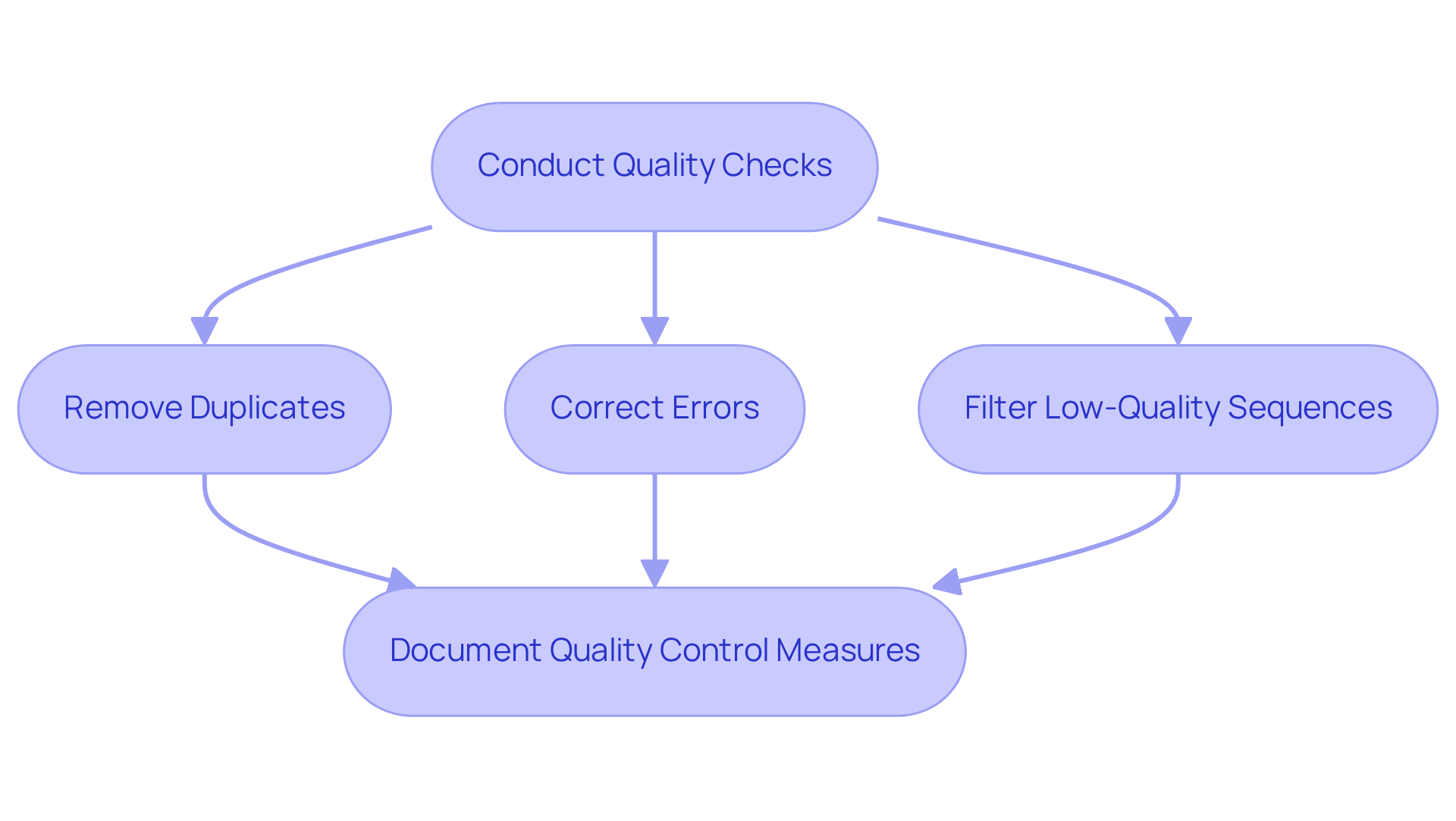
Ensuring the integrity of genomic analysis data is paramount once it is collected. The first step involves conducting thorough quality checks to assess completeness, accuracy, and consistency. Bioinformatics tools, such as FastQC, are essential for evaluating sequencing quality, as they help identify potential issues like low-quality reads and contamination.
Given that each family member possesses a complete electronic genomic health record integrated into TrakGene’s pedigree, maintaining information integrity is vital. This involves:
- Cleaning the data by removing duplicates
- Correcting errors
- Filtering out low-quality sequences
Furthermore, the integrated cancer risk tools enhance patient information by calculating and storing risk scores, underscoring the necessity of high-quality data.
It is also crucial to document all quality control measures undertaken to ensure transparency and reproducibility in the genomic analysis process. This meticulous approach guarantees that the information utilized in subsequent analyses is both reliable and valid.
Process Genomic Data for Analysis
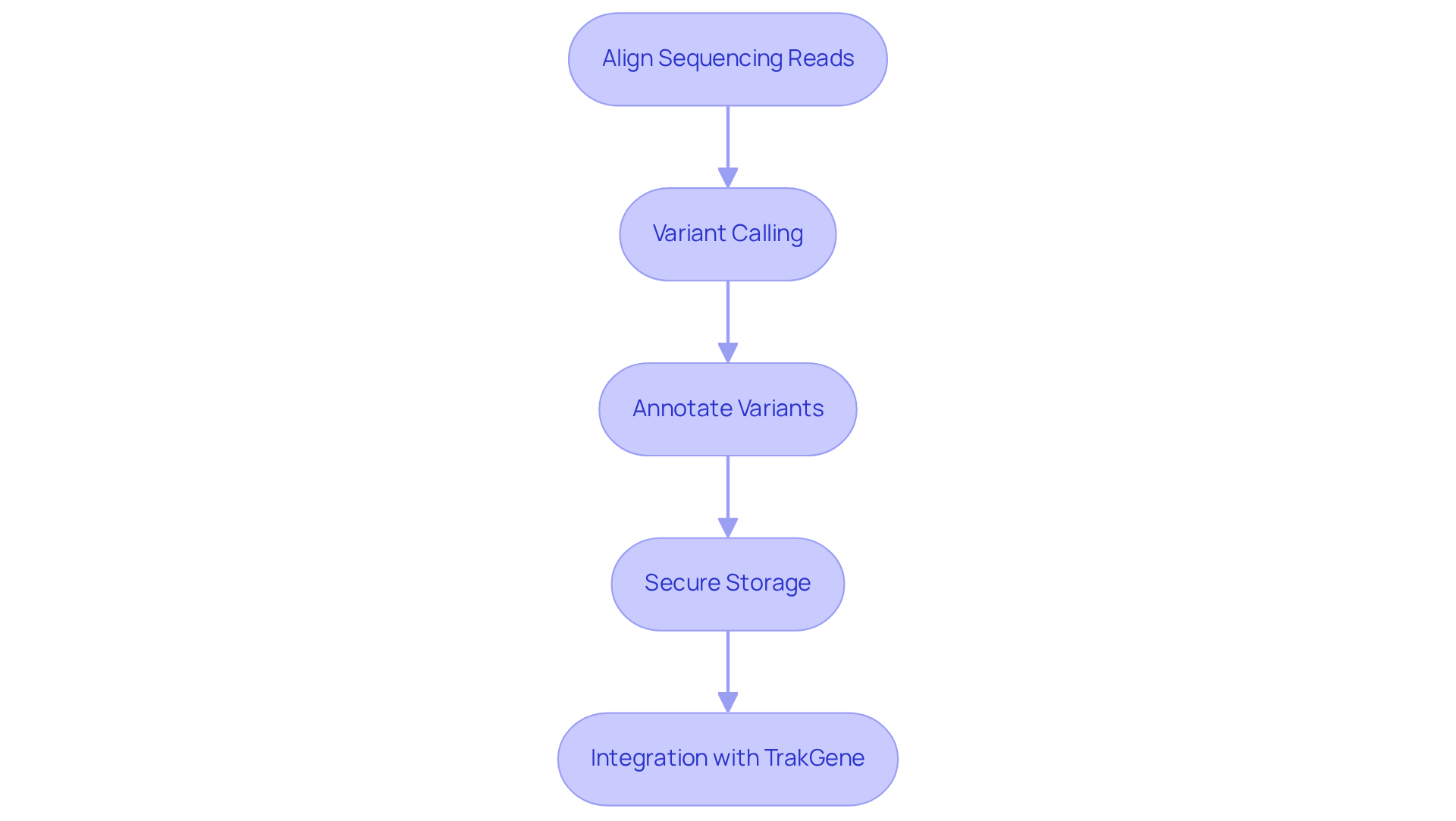
Processing genomic analysis information is a critical step following its cleaning. This process begins with aligning sequencing reads to a reference genome, utilizing efficient tools such as BWA or Bowtie, which are well-regarded for their capability to handle large datasets effectively.
Once the reads are aligned, the next phase of genomic analysis involves variant calling to identify variations, including single nucleotide polymorphisms (SNPs) and insertions/deletions (indels). Industry-standard software like GATK and SAMtools are employed for genomic analysis, providing robust workflows that are optimized for both accuracy and computational efficiency.
After identifying variants, it is essential to annotate them with relevant biological information. This annotation aids in interpretation and supports clinical decision-making. Furthermore, it is crucial to ensure that the processed information is stored securely while remaining easily accessible for future analyses, thereby supporting ongoing research and care for individuals.
TrakGene enhances this process by offering advanced biogenetic management solutions that prioritize security and user access controls. It also provides seamless interoperability with systems via HL7 FHIR. Additionally, TrakGene offers customizable deployment options, allowing genetic counselors to integrate its functionalities into their workflows, ensuring a smooth experience for both management and patient engagement.
Conduct Exploratory Data Analysis and Modeling
To effectively analyze processed genomic information, begin with exploratory analysis aimed at identifying trends, patterns, and anomalies. This foundational step is crucial for understanding the data’s implications. Utilize statistical software such as R or Python to visualize the data through various plots and graphs, which will aid in interpreting complex information.
In this phase, consider employing genomic analysis with clustering analyses to group similar genetic profiles, which can reveal significant insights into genetic relationships. Additionally, regression models may be utilized to assess the relationship between genetic variants and phenotypic traits, providing a deeper understanding of how these factors interact.
Document your findings and hypotheses meticulously, as these insights will not only guide further analysis but also inform clinical recommendations, ensuring that the research translates into practical applications.
Visualize and Report Findings
The final step in the genomic analysis process is to effectively visualize and report findings, particularly through TrakGene’s innovative solutions. Visualization tools such as ggplot2 in R or Matplotlib in Python should be utilized to create clear, informative graphics that emphasize key results.
A comprehensive report must be prepared, encompassing an overview of the analysis process, detailed findings, and their clinical implications. This report should integrate each family member’s electronic genomic health record from TrakGene’s pedigree chart solutions. It is essential to customize the report for the intended audience – whether healthcare providers, individuals seeking care, or researchers – ensuring clarity and relevance.
As Zandra C. Deans states, “As a general rule, each individual should be reported on a separate and unique document,” which underscores the importance of individualized reporting. TrakGene’s integrated cancer risk tools can enhance these reports by calculating and storing risk scores, providing actionable insights that directly improve care for individuals.
Specific suggestions for additional testing or interventions based on the genetic insights obtained should be included. Furthermore, the report must state the date of sample collection and arrival, as this information is crucial for clarity. This step is vital for converting genomic data through genomic analysis into actionable clinical strategies, thereby improving care and decision-making.
Additionally, a clear policy on reporting incidental findings should be established, as this is essential for navigating complex results and effectively communicating them to patients.
Conclusión
Understanding genomic analysis is crucial for genetic counselors, as it underpins personalized healthcare strategies. By leveraging genetic data, counselors can customize care plans to address individual health risks and treatment responses, ultimately improving patient outcomes. This article has presented a comprehensive six-step approach that covers everything from data collection to reporting findings, equipping genetic counselors to navigate the complexities of genomic analysis effectively.
Key insights include the significance of ethical data collection, the necessity for rigorous quality checks, and the application of advanced tools for data processing and visualization. Each step is vital in maintaining data integrity and ensuring that the insights derived from genomic analysis are both accurate and actionable. By integrating these practices, genetic counselors can interpret genetic information effectively and translate it into meaningful clinical recommendations.
The importance of genomic analysis in healthcare is paramount. As the field evolves, genetic counselors must remain proactive in adopting best practices and staying informed about emerging technologies. This commitment not only empowers individuals with knowledge about their genetic predispositions but also promotes a more personalized approach to healthcare, leading to improved health outcomes. Embracing these steps will position genetic counselors as pivotal players in the advancement of precision medicine, making a lasting impact on the lives of those they serve.
Preguntas frecuentes
What is genomic analysis and why is it important?
Genomic analysis involves understanding an individual’s hereditary material, identifying variations that can influence health, disease risk, and treatment responses. It is crucial for creating personalized care plans tailored to each person’s unique genetic profile.
How does genomic analysis benefit genetic counselors?
Genomic analysis allows counselors to assess inherited conditions, guide treatment decisions, and inform individuals about their hereditary risks, enhancing patient care and empowering individuals with knowledge about their genetic predispositions.
What advancements have been made in genomic analysis?
Recent advancements include the shift towards multi-omics approaches, which integrate genetic data with longitudinal clinical records, revealing trends in cancer susceptibility and rare diseases, thus facilitating more accurate risk evaluations.
What is the significance of collecting relevant genomic data?
Collecting extensive genetic information, including DNA samples and relevant clinical information, is crucial for effective genomic analysis, providing context for the genetic data.
What sources can be used to collect genomic data?
DNA samples can be obtained from blood, saliva, or tissue. Additionally, relevant clinical information such as family history and medical records should be gathered.
What ethical guidelines must be followed in genomic data collection?
Information collection practices must adhere to ethical guidelines and regulatory standards, including HIPAA and GDPR, to protect patient privacy and maintain data integrity.
What role does TrakGene play in genomic data compliance?
TrakGene ensures compliance with regulations by establishing an ISO27001 Information Security Management System to uphold industry standards for information security.
What recent legislative efforts impact genetic information practices?
Nebraska’s 2024 law requires direct-to-consumer testing companies to publish privacy policies and obtain consent for information collection. Additionally, the Bipartisan Genomic Information Protection Act (GDPA) aims to regulate hereditary information collection.
Why is it important to be aware of state regulations regarding genetic information?
Variability in state regulations can significantly influence compliance and best practices in the collection of genetic information, making it essential for practitioners to stay informed.
List of Sources
- Define Genomic Analysis and Its Importance
- 2026: A turning point for precision health (https://illumina.com/company/news-center/feature-articles/2026–a-turning-point-for-precision-health.html)
- Emerging Health Innovations In 2026 – Personalized Medicine And Precision Care – North American Community Hub (https://nchstats.com/health-innovations-personalized-medicine)
- AI in Precision Medicine Report 2026-2040: Industry Trends and Global Forecasts – Market to Grow by Over $120 Billion, at a CAGR of 28% (https://finance.yahoo.com/news/ai-precision-medicine-report-2026-090500981.html)
- Keys to Succeed in the Booming $300 Million Optical Genome Mapping Market 2026-2030: Bionano Genomics, 10x Genomics, and Oxford Nanopore Technologies Lead (https://globenewswire.com/news-release/2026/04/13/3272643/28124/en/keys-to-succeed-in-the-booming-300-million-optical-genome-mapping-market-2026-2030-bionano-genomics-10x-genomics-and-oxford-nanopore-technologies-lead.html)
- National Genome Projects are Foundational to Precision Medicine at Scale (https://goldenhelix.com/blog/national-genome-projects-are-foundational-to-precision-medicine-at-scale)
- Collect Relevant Genomic Data
- NIH Seeks Input on New Controlled-Access Data Policy and More (https://about.citiprogram.org/blog/nih-requests-public-input-on-new-controlled-access-data-policy-and-updates-to-genomic-data-sharing)
- Bipartisan Genomic Data Protection Act Reintroduced in Senate (https://hipaajournal.com/genomic-data-protection-act)
- NIH Requests Public Input on New Human Data Protection Policies | COSSA (https://cossa.org/nih-requests-public-input-on-new-human-data-protection-policies)
- Perform Data Quality Checks and Cleaning
- Illumina and the Center for Data-Driven Discovery in Biomedicine bring genomic data and scalable software to the fight against pediatric cancer and rare disease (https://illumina.com/company/news-center/press-releases/press-release-details.html?newsid=a6de4a6d-a68c-44e9-9575-b3dcb0a82838)
- Statistical guidelines for quality control of next-generation sequencing techniques (https://life-science-alliance.org/content/4/11/e202101113)
- How-to: NGS Quality Control – Front Line Genomics (https://frontlinegenomics.com/how-to-ngs-quality-control)
- Standardization and quality management in next-generation sequencing – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC5025460)
- Data Cleaning — Genomics Tutorials 0.1 documentation (https://genomes.readthedocs.io/en/latest/data_cleaning.html)
- Process Genomic Data for Analysis
- Dayhoff Health, AMD Accelerate Genomic Analysis by up to 330x (https://amd.com/en/blogs/2026/dayhoff-health-amd-accelerate-genomics-analysis-by-up-to-330x.html)
- GATK (https://gatk.broadinstitute.org/hc/en-us)
- Accelerating Genomic Data Analysis with an Automated Mutation Calling Solution (https://medium.com/@sequoiaat/accelerating-genomic-data-analysis-with-an-automated-mutation-calling-solution-b16acea6bdf9)
- Benchmarking Variant Calling Tools – Wong et al., 2025 (https://news.varsome.com/en/benchmarking-variant-calling-tools)
- Visualize and Report Findings
- Recommendations for reporting results of diagnostic genomic testing – European Journal of Human Genetics (https://nature.com/articles/s41431-022-01091-0)
- NIH’s All of Us Research Program returns genetic health-related results to participants (https://nih.gov/news-events/news-releases/nih-s-all-us-research-program-returns-genetic-health-related-results-participants)
- Proactive visual and statistical analysis of genomic data in Epiviz – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7868663)
- New guidelines on how to report clinical sequencing data help doctors make decisions about cancer treatment (https://nationaltribune.com.au/new-guidelines-on-how-to-report-clinical-sequencing-data-help-doctors-make-decisions-about-cancer-treatment)






