Introduction
Genomic sequencing is a cornerstone of modern medicine, revealing the intricate DNA patterns that shape individual health. This advanced technology not only identifies hereditary mutations associated with various diseases but also enables the development of personalized treatment strategies that significantly improve patient outcomes. As the field progresses with rapid advancements and a variety of methodologies, healthcare professionals must effectively navigate the complexities of genomic sequencing to fully leverage its benefits.
Define Genomic Sequencing and Its Importance
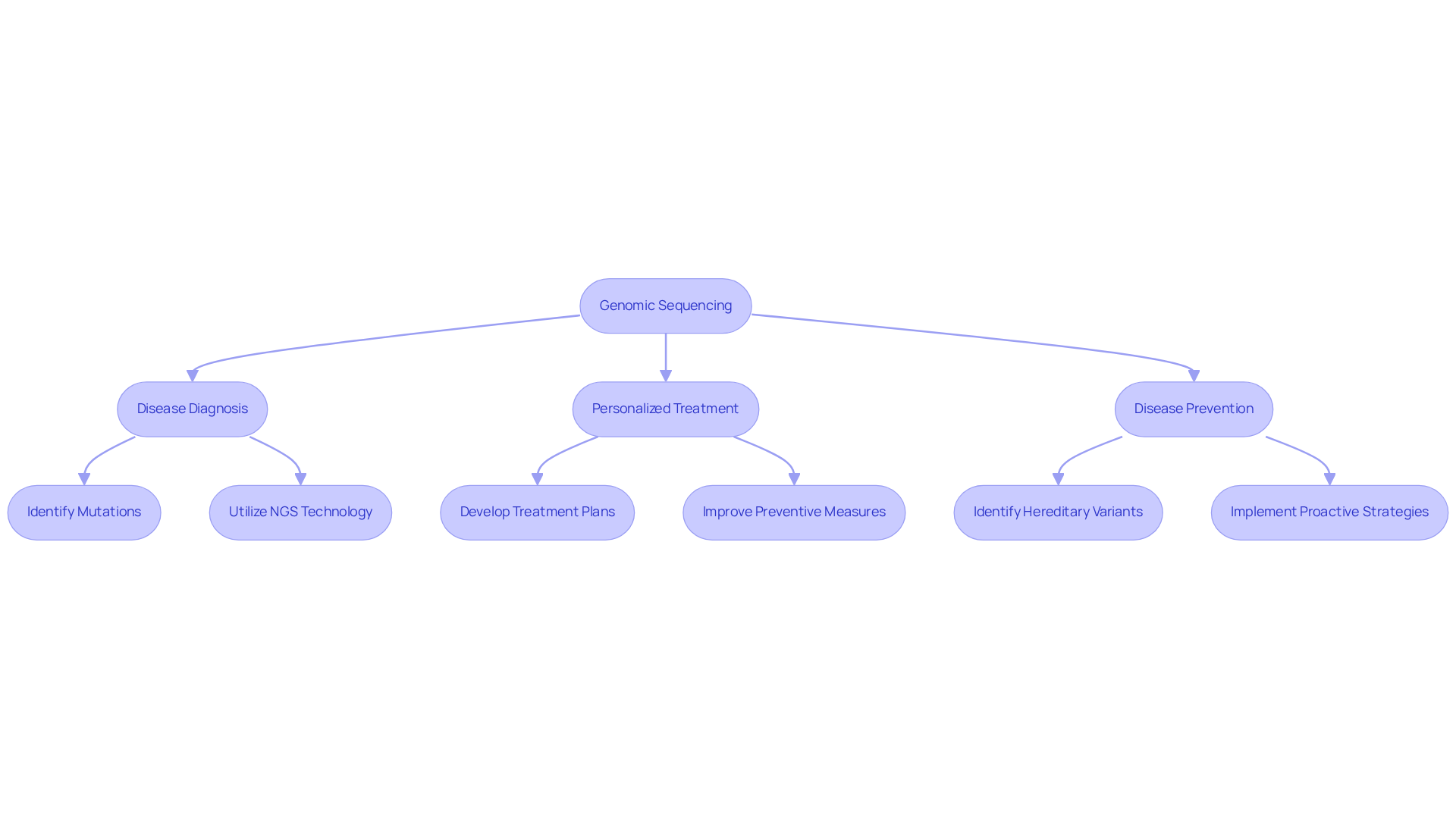
Genomic sequence analysis represents a comprehensive method for determining the complete DNA sequence of an organism’s genome, which includes both chromosomal and mitochondrial DNA. This technology is essential for uncovering hereditary variations that may lead to diseases, allowing healthcare professionals to identify mutations associated with inherited conditions. By decoding the genetic blueprint, practitioners can develop personalized treatment plans and improve preventive measures, thereby significantly advancing the field of precision medicine.
Recent advancements in DNA analysis technology, particularly next-generation sequencing (NGS), have revolutionized disease diagnosis. NGS provides the highest probability of diagnosing rare diseases due to its capability to sequence multiple genes simultaneously. The cost of DNA analysis has decreased dramatically, with projections indicating a reduction of over 90% in recent years, making it increasingly accessible for clinical applications. For instance, the integration of whole-genome analysis into clinical care has improved diagnostic precision, leading to more effective treatment options. As noted by Anna González-Neira, “The data generated will help to determine the risk of each person to develop genetically based diseases, such as cancer.”
Furthermore, the genomic sequence is crucial for disease prevention. By identifying hereditary variants linked to health risks, healthcare professionals can implement proactive strategies tailored to each individual. This approach not only enhances patient outcomes but also improves the overall efficiency of healthcare systems. TrakGene’s automated Pedigree Chart Maker simplifies this process by electronically capturing family history and genetic information, facilitating better analysis and management of health records. Specialists emphasize that the insights gained from DNA analysis can assist in determining an individual’s likelihood of developing hereditary conditions, such as cancer, thus enabling early intervention strategies.
In conclusion, the impact of DNA analysis on healthcare is profound, offering significant advantages such as enhanced diagnostic precision, personalized treatment strategies, and improved preventive care. As the field continues to evolve, integrating genetic data into clinical practice, supported by tools like TrakGene’s pedigree chart solutions, will be vital for advancing patient care and outcomes.
Explore Different Methods of Genomic Sequencing
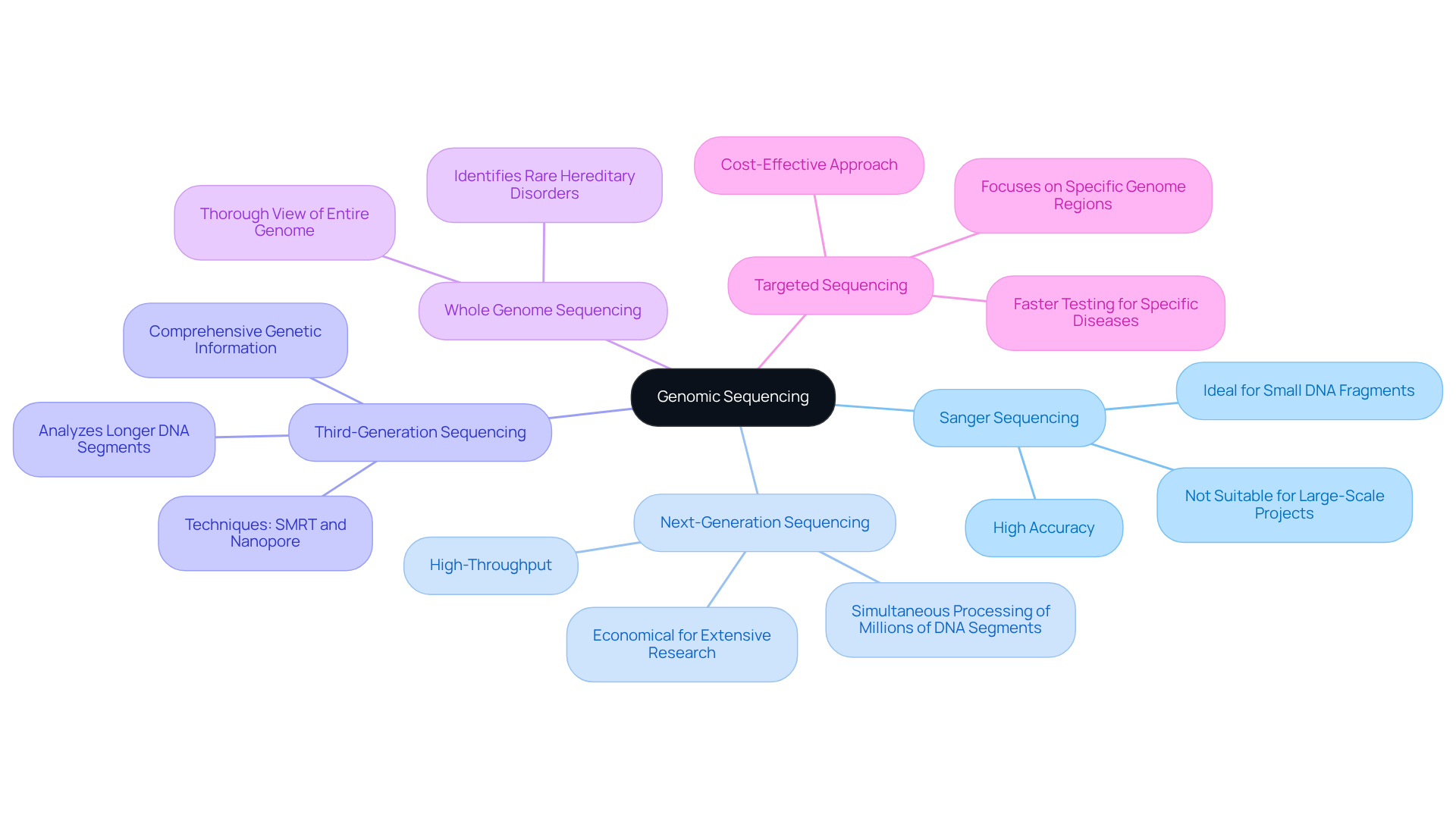
Genomic sequencing encompasses several methods, each offering distinct advantages:
- Sanger Sequencing: This first-generation technique excels in analyzing small DNA fragments. While it boasts high accuracy, it is not ideal for large-scale projects.
- Next-Generation Sequencing (NGS): A high-throughput technique, NGS allows for the simultaneous processing of millions of DNA segments, making it both economical and effective for extensive genetic research.
- Third-Generation Sequencing: Techniques such as single-molecule real-time (SMRT) analysis and nanopore technology facilitate the analysis of longer DNA segments, providing more comprehensive genetic information.
- Whole Genome Sequencing (WGS): This method analyzes the genomic sequence of an individual’s entire genome, offering a thorough view of their hereditary composition, which is crucial for identifying rare hereditary disorders.
- Targeted Sequencing: This approach focuses on specific regions of the genome, such as exomes or panels of genes linked to particular diseases, enabling faster and more cost-effective testing.
Each method serves specific applications, and the selection of a technique typically hinges on the clinical question at hand.
Walk Through the Genomic Sequencing Process
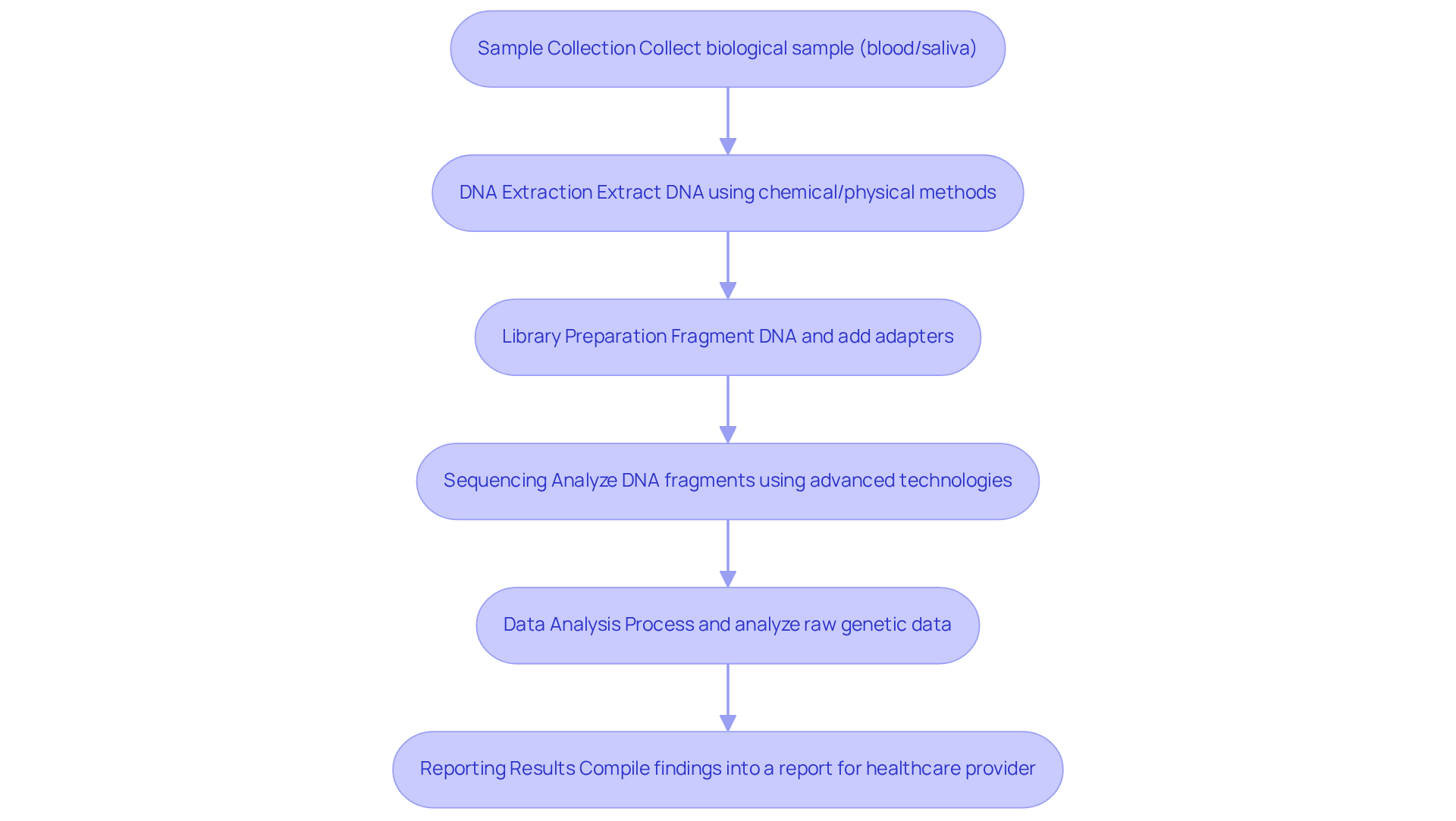
The genomic sequencing process involves several essential steps crucial for the accurate analysis and interpretation of genetic information, particularly in light of advancements highlighted by The 100,000 Genomes Project:
-
Sample Collection: A biological sample, such as blood or saliva, is collected from the patient. Approximately 40% of neonatal deaths are linked to rare hereditary disorders, emphasizing the importance of effective sample collection methods in genomic testing. Insights from Dr. Julian Barwell highlight the necessity for comprehensive data collection to enhance digital pedigrees and improve precision medicine.
-
DNA Extraction: The DNA is extracted from the cells in the sample using various chemical or physical methods. Innovations in extraction techniques, such as on-bead tagmentation, have streamlined this process, eliminating intermediate purification steps while preserving the integrity of the DNA. This method retains the ability to identify copy number variations (CNVs) and structural variants (SVs), which are vital for understanding the genomic sequence related to genetic conditions.
-
Library Preparation: The extracted DNA is fragmented, and specific adapters are added to the ends of the fragments to facilitate the reading of genetic information. Library preparation typically takes an average of 45 minutes from purified genomic DNA and 72 minutes from blood samples, enhancing workflow efficiency.
-
Sequencing: The prepared DNA library is loaded onto a platform, where the DNA fragments are analyzed using advanced technologies. The data yield is approximately 150 Gb at over 40× coverage, with 87% of reads achieving high-quality scores. The average duration for the runs is noted at 11 hours and 12 minutes, contributing to the overall efficiency of the process.
-
Data Analysis: The raw genetic data is processed and analyzed using bioinformatics tools to identify genomic sequences and interpret their potential health implications. The average time for primary and secondary analysis has been significantly reduced to 47 minutes with the latest tools, improving turnaround times for clinicians. This efficiency is crucial, especially considering that rapid whole-genome sequencing (rWGS) can provide fast and accurate diagnoses in critically ill infants, with a positive diagnosis rate of up to 57% in clinical studies. The results from The 100,000 Genomes Project further support the integration of genomic sequence data into clinical practice, enhancing care for individuals.
-
Reporting Results: The findings are compiled into a report that is shared with the healthcare provider, who can then discuss the results with the individual and recommend further action if necessary. Genetic advisors play a vital role in this step, ensuring that individuals understand the implications of their results, particularly in light of advancements in precision medicine stemming from large-scale DNA studies.
Understanding this workflow is essential for both clinicians and individuals, as it underscores the complexity and accuracy involved in genomic testing, ultimately leading to improved health outcomes.
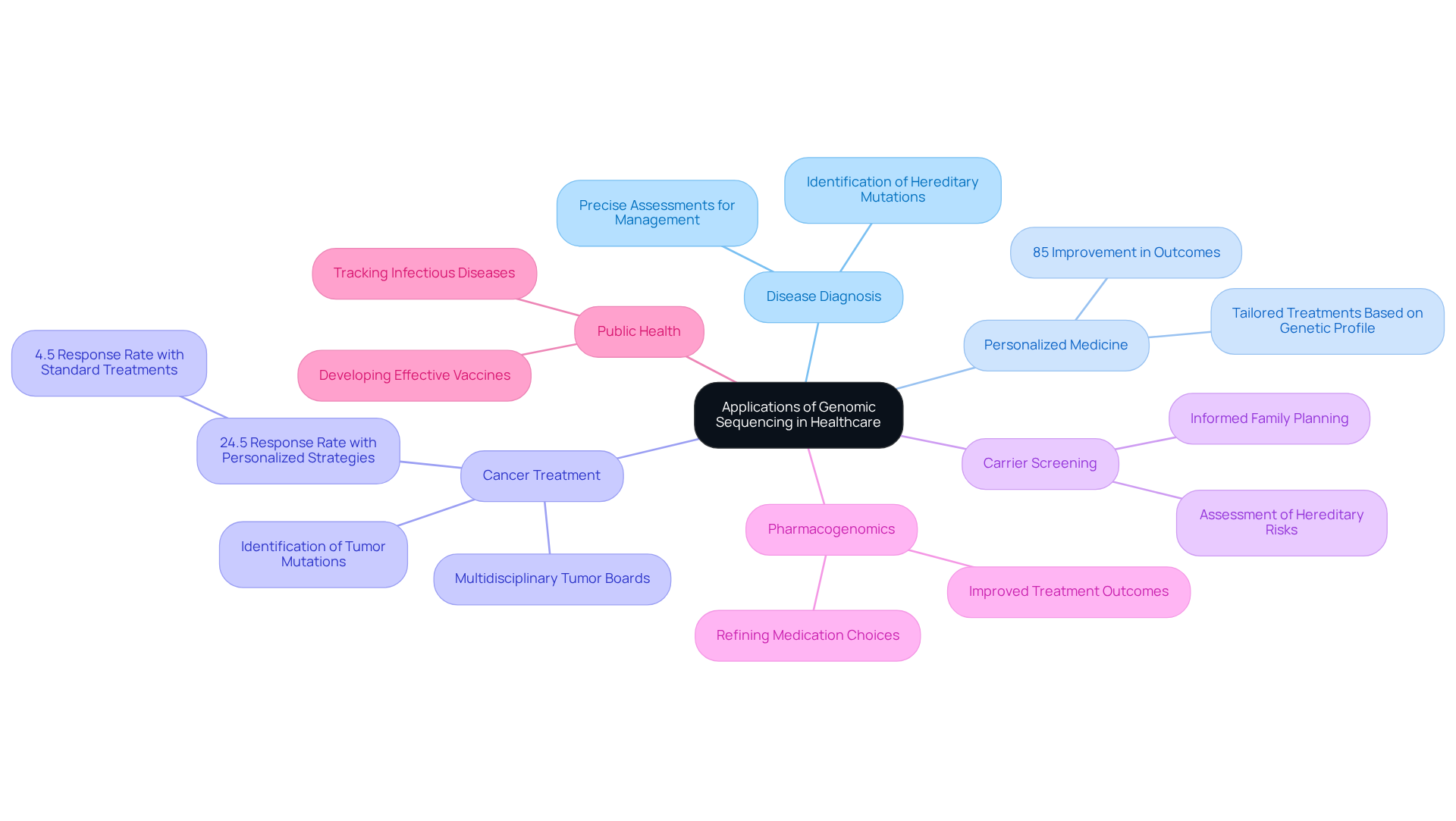
Understand the Applications of Genomic Sequencing in Healthcare
Genomic sequencing has fundamentally transformed healthcare, showcasing its diverse applications:
-
Disease Diagnosis: This technology facilitates the identification of hereditary mutations linked to inherited conditions, enabling precise assessments that significantly impact individual management.
-
Personalized Medicine: By evaluating an individual’s genetic profile, clinicians can tailor treatments, thereby enhancing efficacy and reducing adverse effects. Research indicates that personalized approaches can yield an 85% improvement in outcomes through genomically-matched therapies.
-
Cancer treatment benefits significantly from genomic sequence analysis, which plays a crucial role in identifying specific mutations within tumors and guiding targeted therapies. For example, a study revealed that solid tumors treated with personalized strategies achieved a 24.5% response rate, in contrast to a mere 4.5% with standard treatments. Multidisciplinary tumor boards frequently leverage this data to make informed treatment decisions, ensuring patients receive optimal care.
-
Carrier Screening: This application allows prospective parents to assess their hereditary risks for passing on inherited conditions, fostering informed family planning.
-
Pharmacogenomics: By investigating how genetic variations affect drug responses, pharmacogenomics aids in refining medication choices and dosages, ultimately improving treatment outcomes.
-
Public Health: Genomic sequencing is vital for tracking infectious diseases, comprehending outbreak dynamics, and developing effective vaccines, thereby enhancing public health strategies.
In addition to these applications, TrakGene’s digital solutions streamline pedigree chart creation and health data management, ensuring compliance with HIPAA and GDPR standards. This commitment to data protection, coupled with the advanced pedigree chart tool, underscores TrakGene’s contribution to promoting precision medicine and improving care for individuals. These applications highlight the transformative influence of genomic sequence on contemporary healthcare, reinforcing its essential role in advancing precision medicine and enhancing patient care.
Conclusion
The critical role of genomic sequencing in modern healthcare cannot be overstated. It is essential for healthcare providers to develop a comprehensive understanding of its methods, processes, and applications. By decoding individuals’ genetic makeup, genomic sequencing not only aids in diagnosing diseases but also facilitates personalized medicine, tailoring treatments to the unique genetic profiles of patients.
Key advancements in sequencing technologies, such as next-generation sequencing and third-generation methods, significantly enhance diagnostic accuracy and efficiency. Each step in the genomic sequencing process, from sample collection to data analysis, is vital. This ensures that healthcare professionals can make informed decisions based on robust genetic insights. Furthermore, the various applications of genomic sequencing – including disease diagnosis, pharmacogenomics, and public health – illustrate its transformative impact on patient care and its role in supporting proactive healthcare strategies.
As the field continues to evolve, it is imperative for healthcare providers to embrace genomic sequencing to improve patient outcomes. The integration of advanced sequencing techniques and digital solutions, such as those offered by TrakGene, streamlines workflows and enhances the precision of medical interventions. Engaging with these developments empowers both clinicians and patients to harness the full potential of genomic data, ultimately leading to a healthier future.
Questions fréquemment posées
What is genomic sequencing?
Genomic sequencing is a comprehensive method for determining the complete DNA sequence of an organism’s genome, which includes both chromosomal and mitochondrial DNA.
Why is genomic sequencing important?
It is essential for uncovering hereditary variations that may lead to diseases, allowing healthcare professionals to identify mutations associated with inherited conditions and develop personalized treatment plans.
How has next-generation sequencing (NGS) changed disease diagnosis?
NGS has revolutionized disease diagnosis by providing the highest probability of diagnosing rare diseases through its capability to sequence multiple genes simultaneously.
What has been the impact of cost reduction in DNA analysis?
The cost of DNA analysis has decreased dramatically, with projections indicating a reduction of over 90% in recent years, making it increasingly accessible for clinical applications.
How does genomic sequencing contribute to disease prevention?
By identifying hereditary variants linked to health risks, genomic sequencing allows healthcare professionals to implement proactive strategies tailored to individuals, enhancing patient outcomes and healthcare efficiency.
What role does TrakGene’s automated Pedigree Chart Maker play in genomic analysis?
TrakGene’s automated Pedigree Chart Maker simplifies the process of capturing family history and genetic information electronically, facilitating better analysis and management of health records.
What are the benefits of integrating genetic data into clinical practice?
Integrating genetic data into clinical practice offers enhanced diagnostic precision, personalized treatment strategies, and improved preventive care, ultimately advancing patient care and outcomes.