Introduction
Effective genomic health record management is essential for healthcare organizations, as it lies at the intersection of advanced technology and regulatory compliance. This integration of genomic data into electronic health records is increasingly vital for delivering personalized patient care. By understanding best practices in this domain, organizations can significantly improve clinical outcomes and enhance patient engagement.
Navigating the complexities of regulatory standards poses unique challenges. Organizations must ensure that their staff is adequately trained to utilize advanced tools for risk assessment and family history collection. This training is crucial for maximizing the benefits of genomic data while adhering to compliance requirements.
Establish Compliance with Regulatory Standards
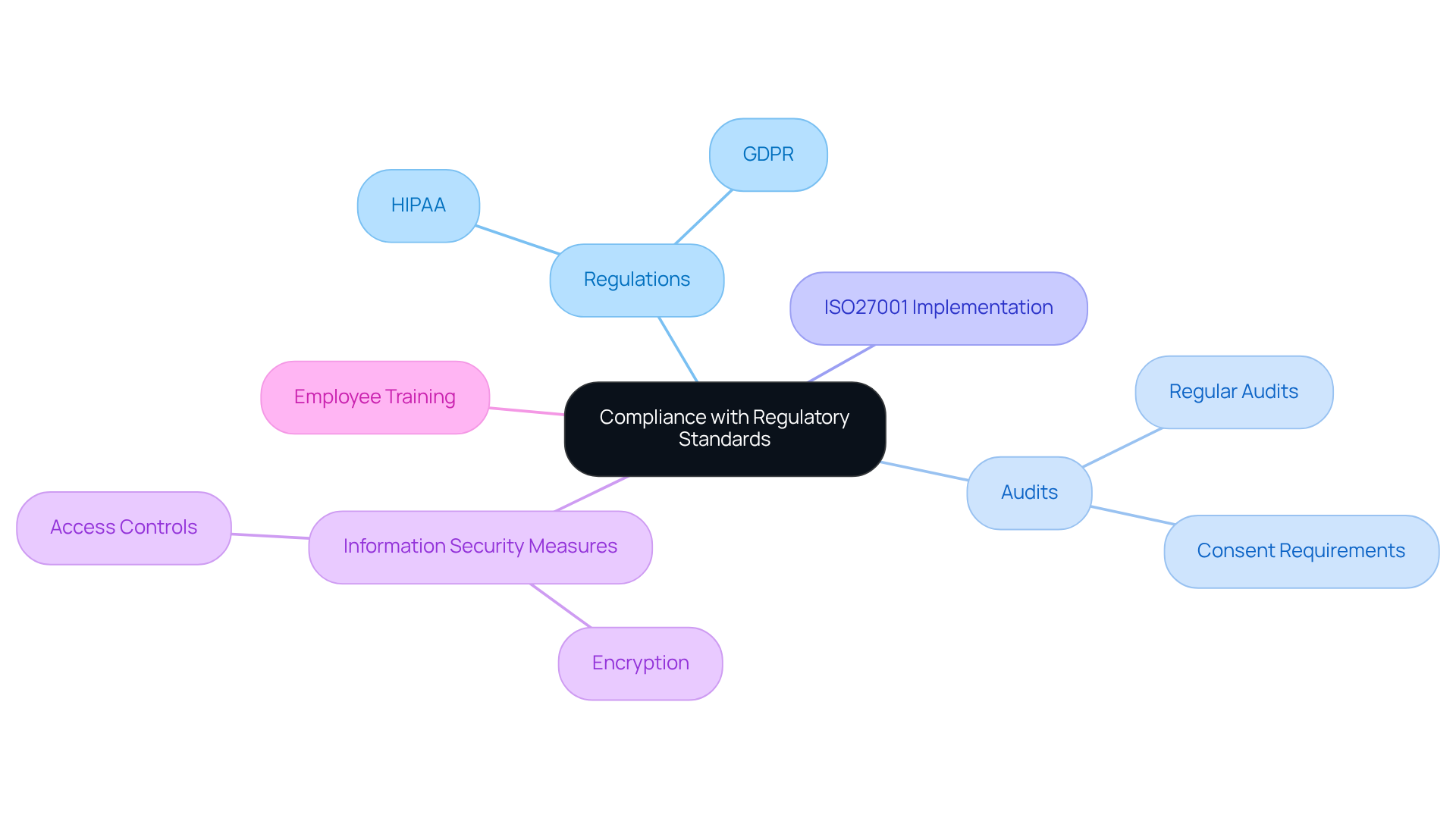
To effectively manage genomic health record management, organizations must prioritize compliance with regulatory standards such as HIPAA and GDPR. TrakGene is committed to this mission, collaborating closely with clients to ensure that all deployments adhere to these essential .
This commitment involves conducting regular audits to verify that all information handling practices meet legal requirements. For instance, GDPR mandates explicit consent for health information processing, a necessity stemming from a final regulation published in 2024 that updated Part 2 while maintaining stricter protections for sensitive SUD details.
To align with industry standards for information security, TrakGene has implemented an ISO27001 Information Security Management System, reinforcing their dedication to safeguarding sensitive genetic information. Employing robust information encryption methods and access controls is crucial in protecting this information from unauthorized access.
Furthermore, organizations should provide training for employees on compliance protocols and the importance of information privacy. By cultivating a culture of adherence and utilizing a checklist of control points to systematize audits, organizations can mitigate risks associated with breaches and enhance their credibility in the eyes of patients and stakeholders.
Integrate Genomic Data into Electronic Health Records
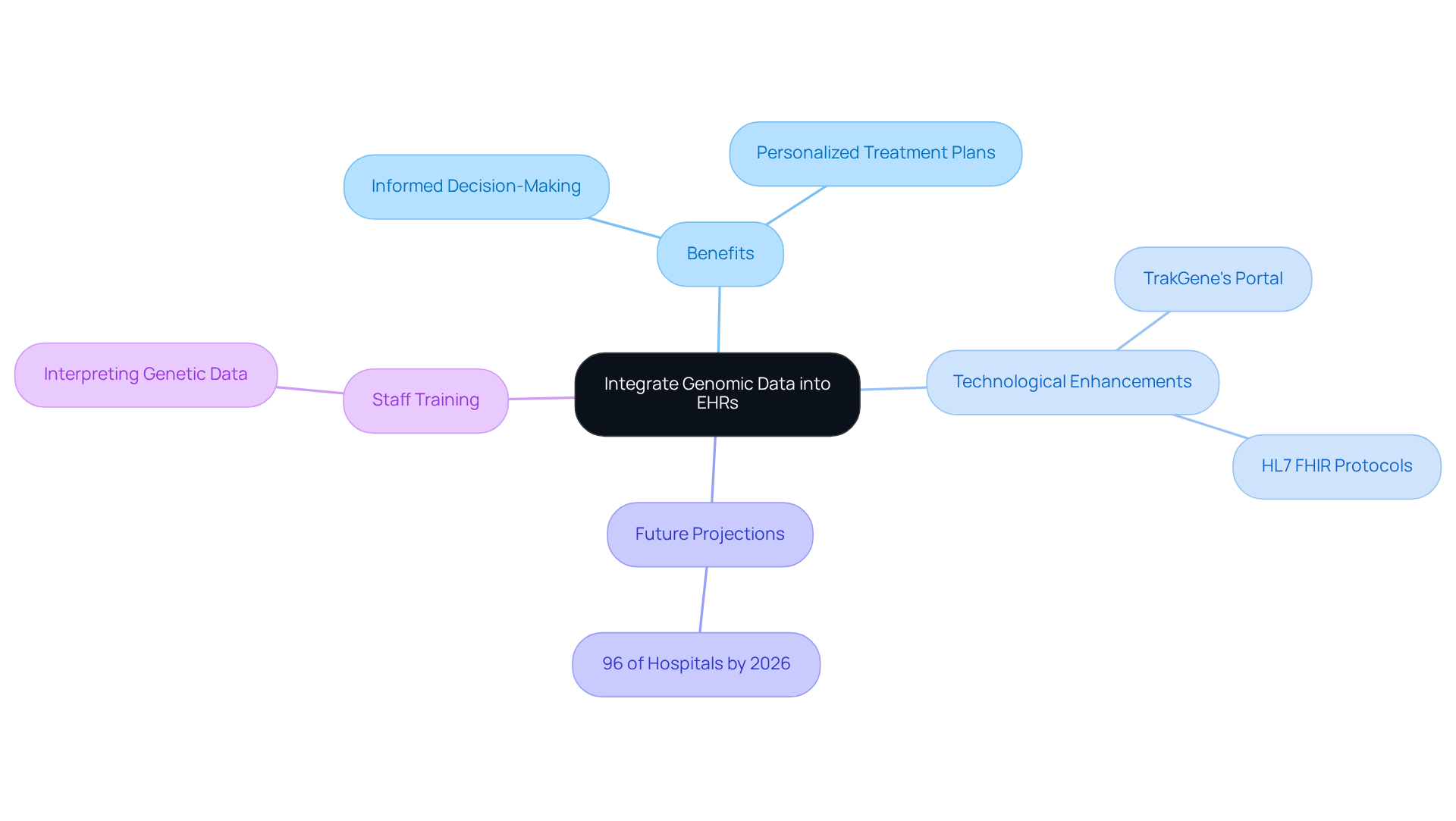
Incorporating DNA data into electronic medical records (EHRs) represents a significant advancement for healthcare providers. This integration enables clinicians to access a patient’s hereditary information alongside their medical history, fostering informed decision-making and personalized treatment plans tailored to each individual’s unique genetic profile.
TrakGene’s enhancements to the Portail d'engagement des patients further facilitate genomic health record management by providing a user-friendly interface for accessing genetic health records. To fully realize the potential of this integration, organizations must adopt standardized information formats. The HL7 FHIR protocols serve as a cornerstone for seamless exchange across various EHR systems.
Looking ahead, the healthcare landscape is projected to experience a substantial increase in EHR integration by 2026, with nearly 96% of hospitals expected to allow patients to view their records electronically. This trend underscores the growing emphasis on interoperability within healthcare.
Moreover, training staff to effectively interpret genetic data is essential for genomic health record management within the EHR context. Such training enhances clinical workflows and empowers healthcare providers to utilize genetic insights for improved patient outcomes. Ultimately, this shift drives the movement towards more .
Utilize Advanced Tools for Family History and Risk Assessment
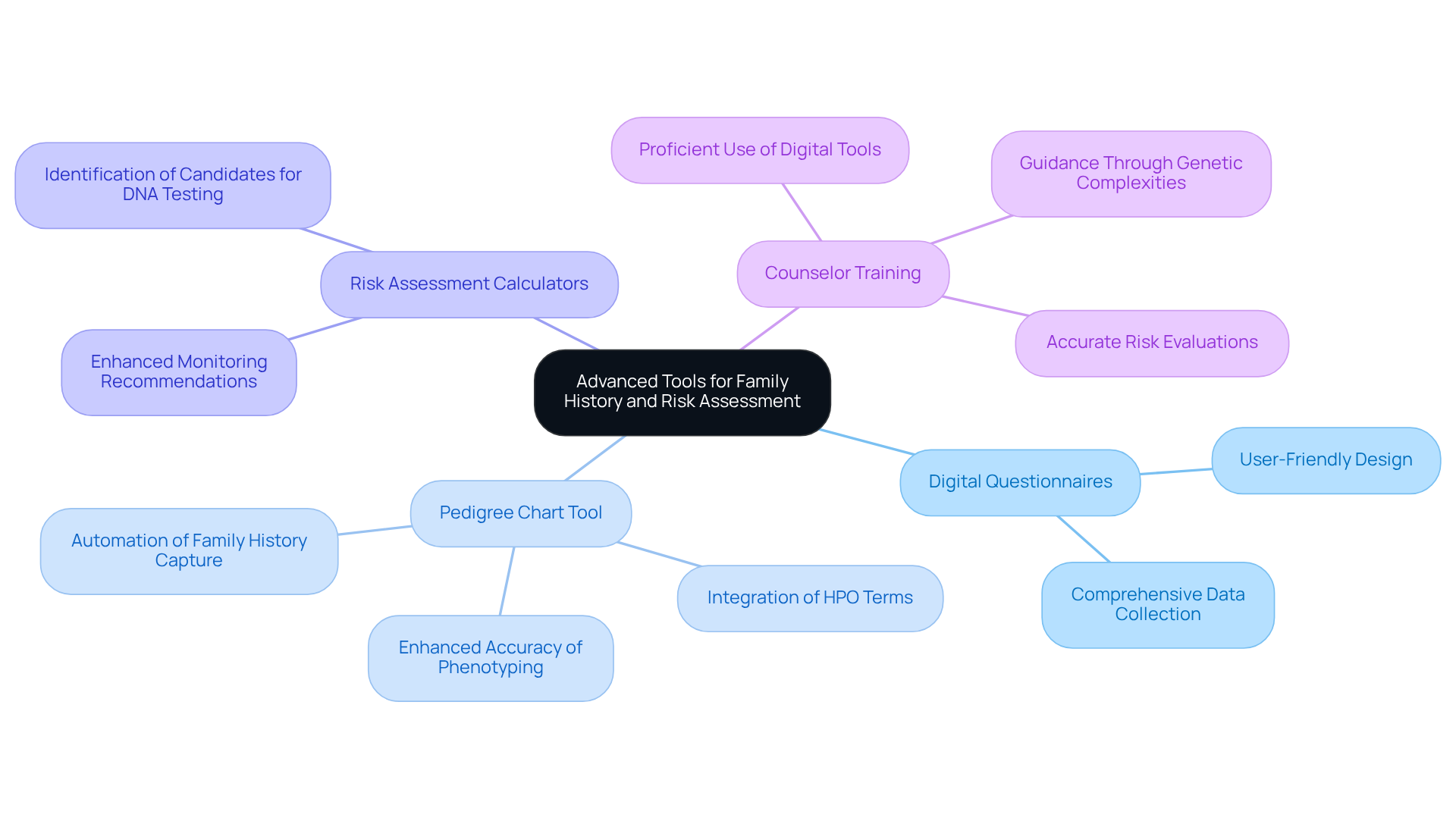
Advanced tools for family history collection and risk evaluation are essential for effective genomic health record management. Organizations must implement user-friendly digital questionnaires that facilitate the comprehensive collection of family medical histories.
For example, TrakGene’s Advanced Pedigree Chart Tool automates the capture of family history, significantly enhancing the accuracy of phenotyping by integrating Ontologie du phénotype humain (HPO) terms. This automation not only streamlines the process but also ensures that the data collected is both precise and relevant.
Additionally, utilizing risk assessment calculators can help identify individuals who may benefit from DNA testing or enhanced monitoring. Insights from counselor Michelle Bowmen underscore the importance of digital tools in cancer outreach, particularly in genomic health record management for effectively managing family medical history.
Training counselors in heredity to is crucial. This training enables them to provide accurate risk evaluations and guide patients through the complexities of genetic information, ultimately improving patient outcomes.
Promote Continuous Education and Training for Genetic Counselors
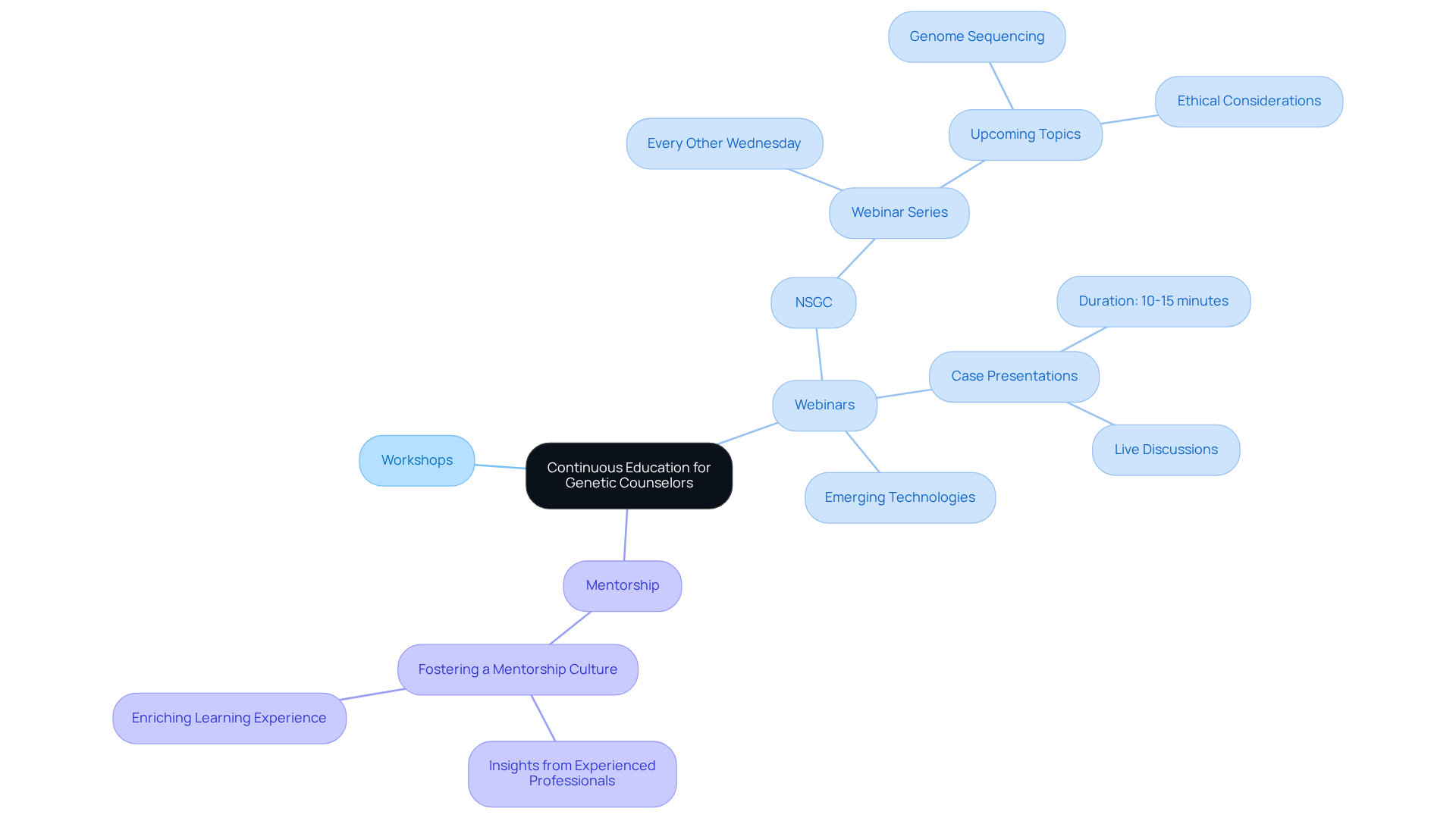
Continuous education and training are essential for counselors to stay informed about advancements in genomic health. Organizations must actively encourage participation in workshops, webinars, and certification programs that focus on the latest developments in genetics and genomics.
For example, the Société nationale des conseillers en génétique (NSGC) provides a series of webinars every other Wednesday. These sessions feature case presentations that enhance practical skills and knowledge. They not only cover but also explore ethical considerations, ensuring counselors are well-prepared to navigate complex scenarios.
Additionally, fostering a mentorship culture within organizations allows new counselors to gain valuable insights from experienced professionals, enriching their learning experience. By prioritizing continuous education, organizations can uphold the highest standards of care for patients facing genetic challenges.
Conclusion
Effective management of genomic health records is essential for advancing personalized healthcare and ensuring compliance with regulatory standards. By prioritizing adherence to laws such as HIPAA and GDPR, organizations can safeguard sensitive genetic information while fostering trust among patients and stakeholders. The integration of genomic data into electronic health records (EHRs) enhances clinical decision-making and paves the way for a more interconnected healthcare landscape.
Key practices for genomic health record management include:
- Establishing compliance frameworks
- Integrating genomic data into EHRs
- Utilizing advanced tools for family history and risk assessment
- Promoting continuous education for genetic counselors
Each of these components is vital in creating a robust system that supports informed patient care and leverages genetic insights for improved outcomes.
As the healthcare industry evolves, the significance of effective genomic health record management cannot be overstated. Organizations are encouraged to embrace these best practices, invest in training and technology, and remain vigilant in compliance efforts. By doing so, they will enhance operational efficiency and contribute to a future where personalized medicine becomes the standard of care, ultimately benefiting patients and the broader healthcare ecosystem.
Questions fréquemment posées
What regulatory standards must organizations comply with for genomic health record management?
Organizations must comply with regulatory standards such as HIPAA and GDPR to effectively manage genomic health record management.
How does TrakGene ensure compliance with these regulatory standards?
TrakGene collaborates closely with clients to ensure that all deployments adhere to essential information protection regulations and conducts regular audits to verify compliance with legal requirements.
What specific requirement does GDPR impose regarding health information processing?
GDPR mandates explicit consent for health information processing, which is essential for compliance.
What recent updates have been made to GDPR regulations?
A final regulation published in 2024 updated Part 2 of GDPR while maintaining stricter protections for sensitive substance use disorder (SUD) details.
What information security measures has TrakGene implemented?
TrakGene has implemented an ISO27001 Information Security Management System and employs robust information encryption methods and access controls to protect sensitive genetic information.
Why is employee training important in compliance with regulatory standards?
Employee training is important to ensure understanding of compliance protocols and the significance of information privacy, which helps cultivate a culture of adherence and mitigates risks associated with breaches.
How can organizations enhance their credibility regarding information protection?
Organizations can enhance their credibility by cultivating a culture of adherence to compliance, utilizing a checklist of control points for audits, and ensuring proper training for employees.