
Introduction
The rapid advancements in genomic medicine present both opportunities and challenges for healthcare organizations. Many organizations struggle to effectively integrate genomic data analysis software into their existing systems. By establishing essential requirements, ensuring data quality, and providing comprehensive training, healthcare providers can unlock the full potential of these powerful tools. To achieve seamless functionality and improved patient outcomes, organizations must navigate the complexities of integration and user support effectively. This article outlines best practices that streamline implementation and enhance the effectiveness of genomic analysis in clinical settings.
Establish Essential Requirements for Software Implementation
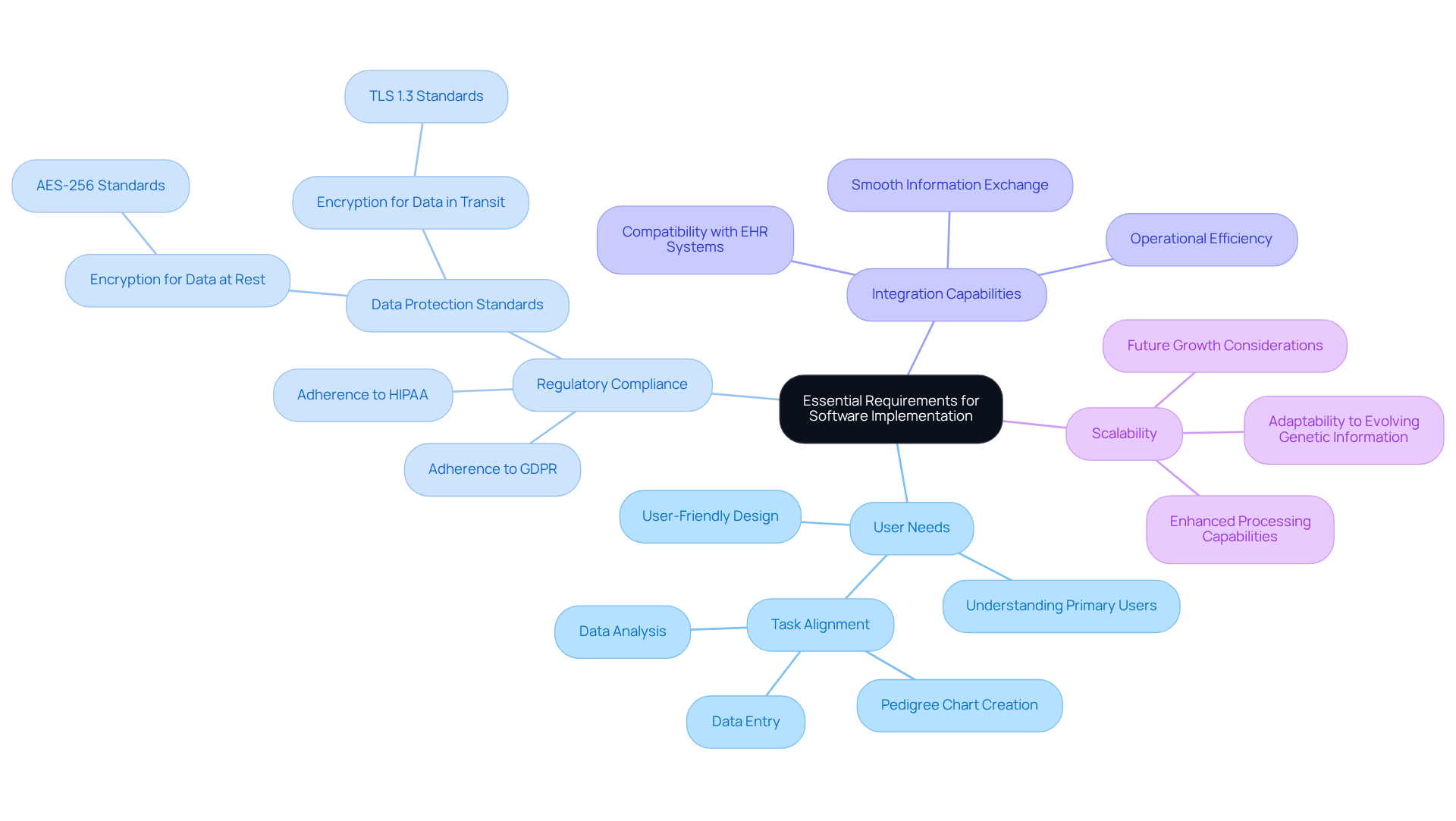
Without a clear understanding of requirements, organizations risk implementing genomic analysis tools that do not meet user needs. To effectively execute genomic analysis tools, organizations must establish clear and comprehensive requirements by involving key stakeholders, including genetic counselors, clinicians, and IT professionals. This collaborative approach ensures that the system effectively addresses their specific needs. Key considerations include:
- User Needs: Understanding the primary users and their tasks-such as pedigree chart creation, data entry, and analysis-is crucial. This focus ensures that the application is user-friendly and aligns with existing workflows.
- Regulatory Compliance: The application must adhere to relevant regulations, including HIPAA and GDPR, to protect patient information and maintain confidentiality standards.
- Integration Capabilities: Assessing the program’s ability to connect with current electronic health records (EHR) and other clinical systems is crucial for enabling smooth information exchange and improving operational efficiency.
- Scalability: Organizations should consider future growth and the potential need for additional features or enhanced processing capabilities, as genetic information continues to evolve.
Ultimately, failing to establish these criteria can hinder effective management of genetic information and limit organizational growth.
Ensure Data Quality and Integrity in Genomic Analysis
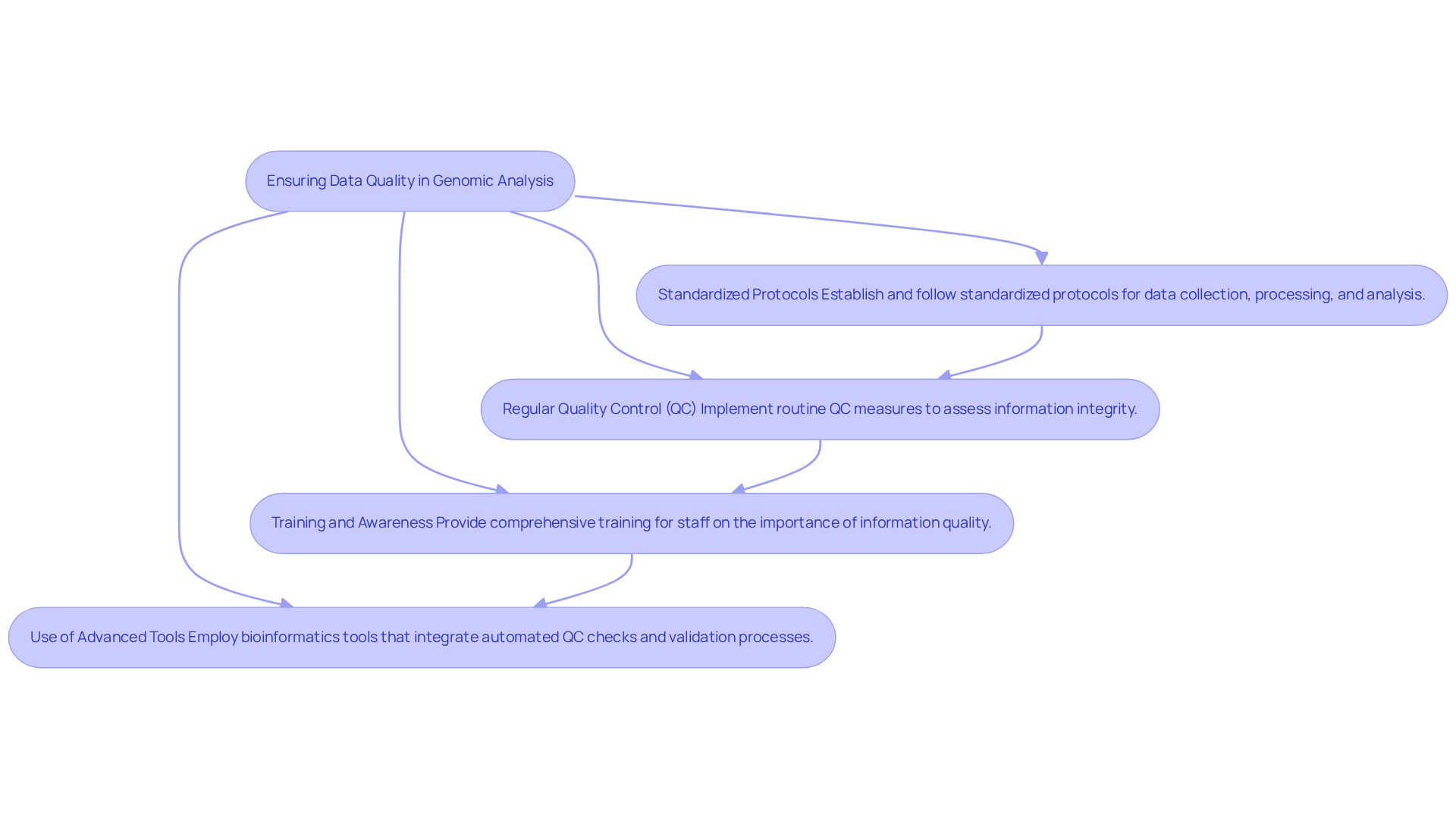
High-quality genomic data analysis software is crucial in genomic analysis, as it directly influences the accuracy of clinical decisions. To ensure high-quality data, organizations should adopt the following best practices:
- Standardized Protocols: Establish and follow standardized protocols for data collection, processing, and analysis. Employing validated tools and methodologies reduces variability and improves consistency in information.
- Regular Quality Control (QC): Implement routine QC measures to assess information integrity. This includes checks for completeness, accuracy, and consistency of genomic information, ensuring that any discrepancies are identified and addressed promptly.
- Training and Awareness: Provide comprehensive training for staff on the importance of information quality and the specific procedures to follow. This fosters a culture of responsibility and diligence in information management, essential for maintaining high standards.
- Use of Advanced Tools: Employ bioinformatics tools that integrate automated QC checks and validation processes. These advanced tools can identify and rectify errors early in the analysis pipeline, further protecting information integrity.
Without high-quality data, the risk of inaccurate clinical decisions increases, potentially jeopardizing patient outcomes. This commitment to data quality translates into more reliable genomic data analysis software, leading to better patient outcomes. Ultimately, the commitment to data quality can significantly enhance patient outcomes and trust in genomic medicine.
Provide Comprehensive Training and Ongoing Support for Users
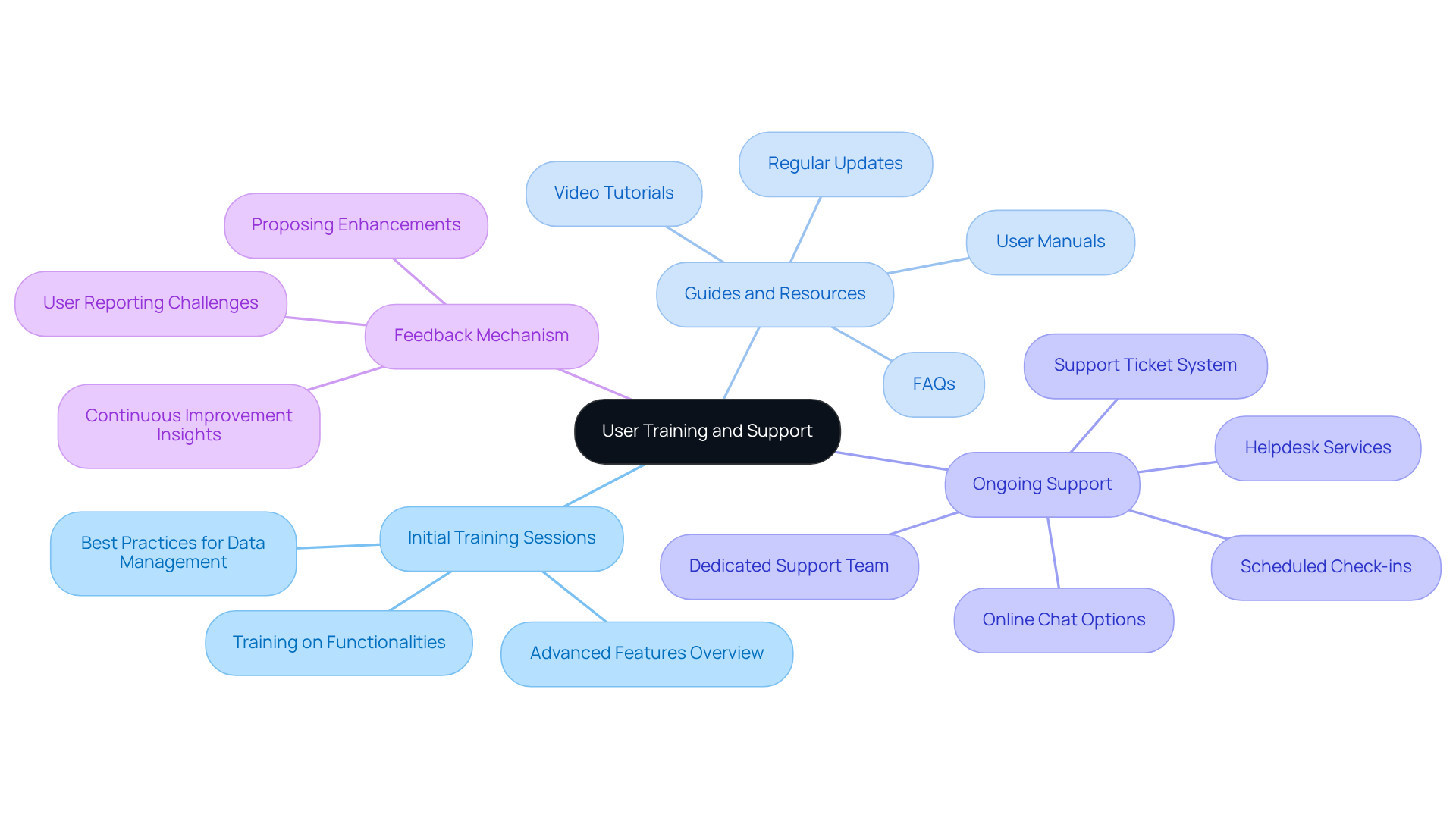
Effective user training and ongoing support are critical for maximizing the benefits of genetic analysis tools.
- Initial Training Sessions: Implement thorough training sessions for all users at the onset of software deployment. These sessions will cover essential functionalities, advanced features, and best practices to ensure effective data management.
- Guides and Resources: Ensure that manuals, video tutorials, and FAQs are readily available through the TrakGene Help Centre, facilitating user access to essential information. Regular updates to these resources are essential to ensure they reflect the latest software enhancements and functionalities.
- Ongoing Support: A dedicated support team should be established to address technical inquiries and issues effectively. This support can take various forms, including helpdesk services, online chat options, or scheduled check-ins to proactively address user concerns. Users can open support tickets, report bugs, and submit requests for new features through the Help Centre, enhancing the overall support experience.
- Feedback Mechanism: Implement a feedback mechanism that allows users to report challenges and propose enhancements, fostering continuous improvement. This method not only enhances satisfaction among individuals but also offers valuable insights for future training initiatives and system updates.
Investing in user training and support is essential for enhancing user proficiency, which consequently results in more effective use of genomic data analysis software and better patient outcomes. Ultimately, a well-structured training and support system can significantly impact patient care and outcomes.
Facilitate Seamless Integration with Existing Healthcare Systems
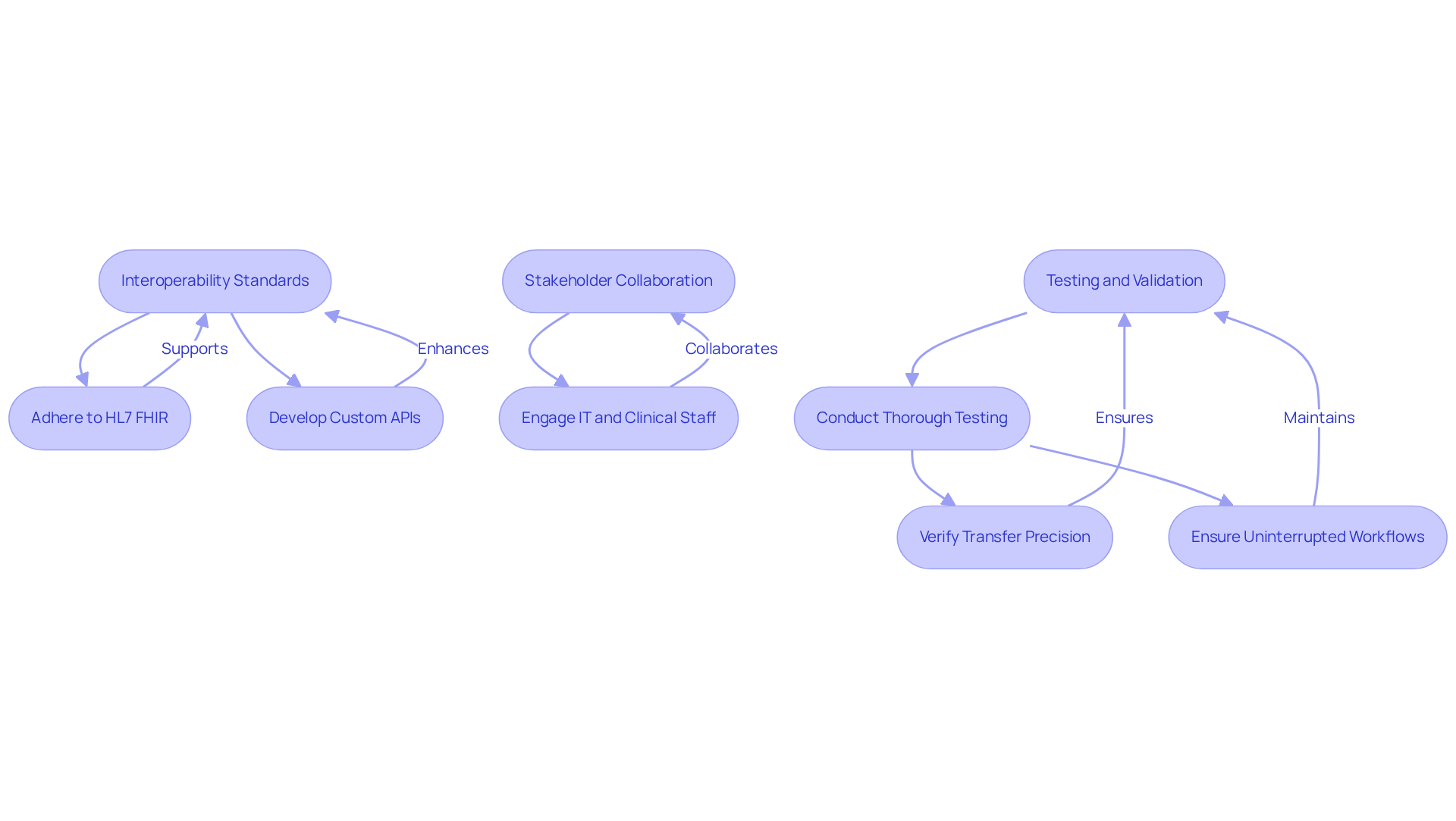
The integration of genomic data analysis software into existing healthcare systems presents significant challenges that require strategic solutions. To achieve seamless integration, organizations should implement the following strategies:
- Interoperability Standards: Adhering to interoperability standards such as HL7 FHIR is essential for facilitating data exchange between diverse systems. The adoption of FHIR is expected to attain 90% of health systems worldwide by 2025, highlighting its importance in incorporating genetic information into electronic health records (EHRs). Developing custom application programming interfaces (APIs) for genomic data analysis software allows for tailored integration with existing systems, enhancing data flow and ensuring that genomic insights are readily accessible to clinicians. This flexibility is crucial for adapting to the unique needs of various healthcare environments.
- Stakeholder Collaboration: Engaging with IT departments, clinical staff, and software vendors is vital for collaboratively designing integration processes. This approach ensures that all perspectives are considered, leading to solutions that effectively meet the needs of all users involved in patient care.
- Testing and Validation: Conducting thorough testing of the integration process is necessary to identify and resolve any issues prior to full deployment. This includes verifying transfer precision and ensuring that workflows remain uninterrupted, which is essential for maintaining operational efficiency.
Ultimately, the success of these strategies will determine the extent to which genomic insights can transform patient care and operational practices.
Conclusion
The integration of genomic data analysis software is essential for advancing patient care and clinical outcomes in today’s healthcare landscape. By focusing on essential requirements, data quality, user training, and seamless integration with existing systems, organizations can create a robust framework that supports the complex needs of genomic analysis. This multifaceted approach ensures that the tools implemented not only meet regulatory standards but also align with user workflows and promote accurate clinical decision-making.
One crucial insight is the need to involve stakeholders in gathering requirements to accurately capture user needs. Additionally, maintaining high data quality through standardized protocols and quality control measures is vital. Comprehensive training and ongoing support for users play a critical role in ensuring effective utilization of these tools. Furthermore, facilitating interoperability and collaboration among healthcare professionals can significantly enhance the integration of genomic insights into patient care.
Ultimately, implementing genomic data analysis software goes beyond technicalities; it represents a genuine commitment to enhancing healthcare outcomes. Organizations are encouraged to prioritize these best practices to foster a culture of excellence in genomic medicine, ensuring that both patients and providers benefit from the transformative potential of genomic data. By embracing these strategies, organizations can not only enhance their practices but also play a pivotal role in the future of healthcare in the genomic era.
Frequently Asked Questions
Why is it important to establish essential requirements for software implementation in genomic analysis?
Establishing essential requirements is crucial to avoid implementing tools that do not meet user needs, ensuring effective execution of genomic analysis tools.
Who should be involved in the requirements establishment process for genomic analysis tools?
Key stakeholders such as genetic counselors, clinicians, and IT professionals should be involved to ensure the system addresses specific user needs.
What are some key considerations when establishing requirements for genomic analysis tools?
Key considerations include understanding user needs, ensuring regulatory compliance, assessing integration capabilities, and considering scalability for future growth.
How does understanding user needs impact the implementation of genomic analysis tools?
Understanding user needs ensures that the application is user-friendly, aligns with existing workflows, and effectively supports tasks like pedigree chart creation, data entry, and analysis.
What regulations must genomic analysis applications comply with to protect patient information?
Applications must adhere to regulations such as HIPAA and GDPR to maintain confidentiality and protect patient information.
Why is it important to assess integration capabilities when implementing genomic analysis tools?
Assessing integration capabilities is crucial for enabling smooth information exchange with current electronic health records (EHR) and other clinical systems, thus improving operational efficiency.
What does scalability refer to in the context of genomic analysis software?
Scalability refers to the software’s ability to grow and accommodate future needs, such as additional features or enhanced processing capabilities as genetic information evolves.
What are the potential consequences of failing to establish clear requirements for genomic analysis tools?
Failing to establish clear requirements can hinder effective management of genetic information and limit organizational growth.
List of Sources
- Establish Essential Requirements for Software Implementation
- Genomic Data Analysis Compliance Requirements Guide (https://lifebit.ai/blog/genomic-data-analysis-compliance-requirements)
- Ensure Data Quality and Integrity in Genomic Analysis
- Data Integrity in Clinical Trials: Best Practices for Ensuring Reliable (https://xceneresearch.com/data-integrity-in-clinical-trials)
- Healthcare Data Quality Challenges in 2026 and How AI Is Solving Them (https://digna.ai/healthcare-data-quality-challenges-2026-ai-solutions)
- WHO releases new principles for ethical human genomic data collection and sharing (https://who.int/news/item/20-11-2024-who-releases-new-principles-for-ethical-human-genomic-data-collection-and-sharing)
- Why data quality is key to AI success in 2026 (https://strategy.com/software/blog/why-data-quality-is-key-to-ai-success-in-2026)
- Provide Comprehensive Training and Ongoing Support for Users
- Bioinformatics: The Importance of Training the Trainer (https://mdibl.org/bioinformatics-the-importance-of-training-the-trainer)
- New research finds genomic professionals support patient genomic data ownership in Australia (https://bioinformatics.csiro.au/blog/new-research-finds-genomic-professionals-support-patient-genomic-data-ownership-in-australia)
- Support of the National Health Genomics Policy Framework and Implementation Plan Review — Australian Genomics (https://australiangenomics.org.au/projects/support-of-the-national-health-genomics-policy-framework-and-implementation-plan-review)
- The Impact of Training on Electronic Health Records Related Knowledge, Practical Competencies, and Staff Satisfaction: A Pre-Post Intervention Study Among Wellness Center Providers in a Primary Health-Care Facility – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC10243608)
- Employee Training Statistics, Trends, And Data In 2025 (https://elearningindustry.com/employee-training-statistics-trends-and-data)
- Facilitate Seamless Integration with Existing Healthcare Systems
- The Future of FHIR: 90% of Health Systems to Adopt FHIR APIs by 2025 (https://ottehr.com/post/health-fhir-apis-2025)
- Ethical data sharing is key to genomic healthcare | Melbourne Genomics (https://melbournegenomics.org.au/guide-genomics/genomic-data/ethical-data-sharing-key-genomic-healthcare)
- Genomics on FHIR – a feasibility study to support a National Strategy for Genomic Medicine – npj Genomic Medicine (https://nature.com/articles/s41525-025-00516-1)
- Global FHIR Adoption Statistics: A Comprehensive Overview – Co-Desion (https://codesion.com/global-fhir-adoption-statistics-a-comprehensive-overview)
- The Importance of Healthcare APIs and Making Data More Secure (https://elationhealth.com/resources/blogs/the-importance-of-healthcare-apis)






